Picture a massive textile dyeing factory that pumps out thousands of liters of brightly colored wastewater, loaded with dyes, chemical oxygen demand (COD), and salt. In some textile plants, the wastewater can contain as much as 5–6% sodium chloride (NaCl), roughly the same concentration as seawater. Now imagine trying to clean that water biologically, using living microorganisms, when salt is one of the most effective ways to kill or suppress microbial life.
That is precisely the challenge researchers tackled in a landmark 2023 study published in the Journal of Environmental Management, and the results are not just fascinating from a scientific standpoint, they carry immediate, practical lessons for environmental engineers, wastewater plant operators, and anyone working at the intersection of biotechnology and industrial sustainability.
The Problem: Textile Wastewater Is a Nasty Beast
The global textile industry is enormous and is one of the most water-intensive manufacturing sectors in the world, with over 90% of the water it uses ending up as wastewater. This effluent is no ordinary dirty water, it is a cocktail of synthetic dyes, heavy metals, organic compounds, and, critically, salt.
Salt is used extensively in the textile dyeing process to help dyes bond to fabric fibers. The result is that effluent from textile plants often carries high salinity, a property that causes serious problems for biological wastewater treatment systems.
Why does salinity matter?
Biological treatment systems rely on living microorganisms to break down pollutants. These microbes, namely, bacteria, archaea, and others, are sensitive to their environment. When salt concentration rises, it creates a high osmotic pressure difference across the cell membrane and allows water to rush out of the cell. The microorganism dehydrates from the inside, loses metabolic function, and can die. This is why salting meat preserves it: it kills the bacteria that would otherwise cause decay.
For a wastewater treatment plant, this means high salinity can devastate the biological communities doing the heavy lifting of purification. The question researchers asked: just how bad is it, and is there a way to predict and manage the damage?
Anaerobic Membrane Bioreactors (AnMBR)
What Is an AnMBR?
An AnMBR combines two powerful water treatment technologies into one system. The first is anaerobic biological treatment, where microorganisms break down organic pollutants without oxygen. This is the same basic process that happens in a compost pile or a cow’s stomach — microbes consume organic matter and produce biogas (mostly methane and CO2) as a byproduct. The second is membrane filtration, where a physical membrane acts as an ultra-fine filter, capturing solids, bacteria, and even fine particles from the water before it leaves the system.
Together, these create a system that can achieve high removal of color, organic load, and suspended solids from complex industrial wastewater, while also recovering energy in the form of biogas. For textile wastewater, AnMBRs are particularly attractive because azo dyes (the most common type of textile dye) are difficult to treat aerobically but break down reasonably well under anaerobic conditions.
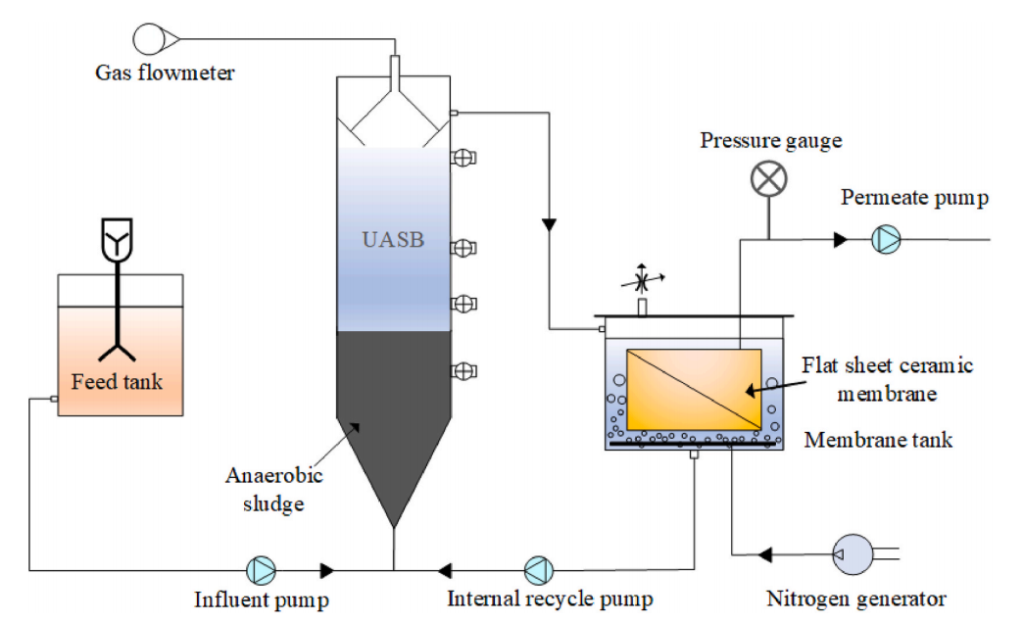
The Specific Setup: UASB + Ceramic Membrane
In this study, the researchers used a specific AnMBR configuration that consists of biological reactor, which was an Upflow Anaerobic Sludge Blanket (UASB) reactor, a technology originally developed in the Netherlands and now widely used around the world. In a UASB, wastewater flows upward through a dense blanket of anaerobic sludge granules, where microorganisms devour the organic compounds.
The membrane used was a flat-sheet alumina (Al2O3) ceramic membrane with a pore size of 0.1 micrometers. Ceramic membranes are more robust than polymer membranes as they can handle higher temperatures, harsh chemicals, and aggressive cleaning protocols better. The ceramic membrane allowed treated water (permeate) to pass through while retaining microorganisms and suspended solids inside the system.
Real-World Setup at a Glance
UASB reactor volume: 6.4 liters | Membrane area: 0.065 m² | Temperature: 35°C (typical mesophilic anaerobic conditions) | Hydraulic Retention Time: ~21 hours | Synthetic textile wastewater: 1,000 mg/L glucose + 100 mg/L methyl orange dye | Salinity tested: 0, 5, 10, and 20 g/L NaCl across four phases lasting 20–30 days each
The Experiment: Turning Up the Salt, Phase by Phase
The researchers ran their AnMBR for about 81 days total, divided into four operational phases. In each phase, they increased the salt concentration in the feed water while keeping everything else as constant as possible.
Think of it like a laboratory stress test. They were asking: at what point does the system start to crack? And what happens inside the biological community as the pressure builds?
1. Phase I — No Salt (Baseline)
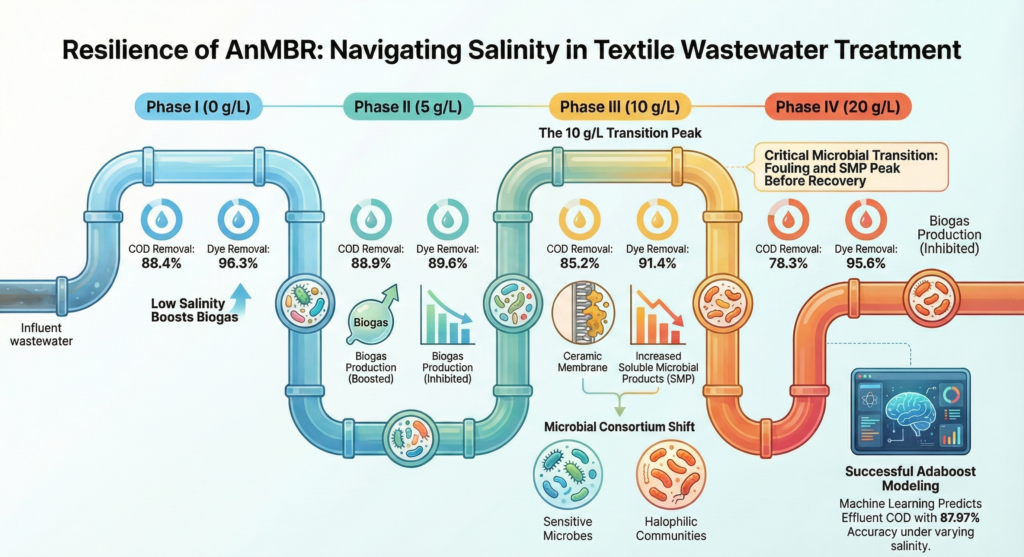
The system operated without any added salt to establish a baseline for performance measurements. The removal of chemical oxygen demand (COD), which indicates the degradation of organic matter, achieved an impressive rate of 88.4%. Even more remarkable was the dye removal rate, surpassing 96%. The microbial community involved was both diverse and flourishing, showcasing a healthy ecosystem.
2. Phase II — 5 g/L NaCl (Low Salinity)
The first dose of salt caused an immediate dip in COD removal, which dropped to about 80.5%. But here is something counterintuitive: it then recovered. By the end of Phase II, the system had bounced back to nearly 88.9% COD removal. This shows the adaptability of microbes. Even more surprisingly, biogas production actually increased in this phase, with the specific methanogenic activity (a measure of methane-producing microbial activity) rising from 0.046 to 0.077 g COD/g VSS/day. This finding challenges the assumption that any salt is bad. Low concentrations of salt appear to act as a manageable stressor that stimulates certain microbial responses without overwhelming the community.
3. Phase III — 10 g/L NaCl (The Crisis Point)
This is where things got interesting and troubling. COD removal settled at 85.2%, but behind that still-acceptable number, the system was under severe internal stress. The membrane began fouling at a dramatically accelerated rate while the concentration of soluble microbial products (SMP), essentially the waste secretions of stressed microbes, spiked to a peak of 178 mg/L. This is where Microbial diversity collapsed, and key species disappeared. The biological community was in the middle of a wholesale transition. Phase III, the researchers concluded, was a transition phase, a turbulent middle ground between the old community adapted to low salinity and a new one capable of surviving in high-salt conditions.
4. Phase IV — 20 g/L NaCl (The New Equilibrium)
A remarkable development has occurred within the system. Despite an increase in salt levels, the system has demonstrated stabilization. Notably, membrane fouling rates have decreased significantly, and soluble microbial products (SMP) concentrations have reverted to approximately 133 mg/L, which is on par with the levels observed during Phase I.
Additionally, although chemical oxygen demand (COD) removal has declined further to 78.3%, this still represents a substantial reduction from the baseline. Encouragingly, the system is now operating with predictable and stable performance. The establishment of a new, salt-tolerant microbial community has contributed positively to these results, facilitating continued effective functioning.
The Microbiology: Who Survived and Why
One of the most valuable parts of this research is the detailed microbial analysis. The researchers used high-throughput DNA sequencing to identify which microorganisms were present at the end of each phase. What they found tells a compelling story of ecological succession under stress.
The Casualties: Salt-Sensitive Species
Several microorganisms that thrived in the absence of salt were essentially eliminated as salinity climbed above 10 g/L:
- Aminiphilus: It is responsible for degrading amino acids, a critical step in breaking down complex organic matter. Its relative abundance dropped from 6–7% all the way to undetectable levels by Phase III.
- Mesotoga: It is a complex carbohydrate degrader. Its decline helps explain the reduced COD removal at high salinity.
- Caldatribacterium: This is involved in acidogenesis (the acid-producing stage of anaerobic digestion).
- Methanobrevibacter, Methanosaeta, and Methanobacterium: the methanogens, the methane-producing archaea that are central to biogas recovery. These actually increased slightly in Phase II (low salt) but crashed below 0.1% relative abundance in Phases III and IV.
The loss of methanogens is particularly significant. These microorganisms are often the most sensitive link in the anaerobic digestion chain, and their suppression explains why biogas production dropped sharply at high salinity.
The Survivors: Salt-Tolerant Species
As the salt-sensitive species died off, a new cast of salt-tolerant microorganisms rose to fill the ecological niches:
- Longilinea: A strict anaerobe that degrades carbohydrates and produces volatile fatty acids. Its relative abundance exploded from 0.3% at zero salinity to 22.4% at 10 g/L. Crucially, it is also involved in aromatic compound degradation, helping explain why dye removal remained relatively stable.
- Ignavibacterium: This is associated with benzene ring opening, relevant for breaking down the aromatic structures in azo dyes.
- Bosea: It is an azo dye degrader. Its increased presence in high-salinity phases directly supports the maintenance of dye removal performance.
- Rhodovarius and Flexilinea: These are both salt-tolerant organisms that helped maintain some level of organic degradation.
“The Ecological Insight
This microbial community transition is essentially natural selection playing out in a controlled reactor. The salt acts as a selection pressure, eliminating organisms that cannot cope with osmotic stress and creating space for halophilic (salt-loving) or halotolerant (salt-tolerating) species. Phase III was the chaotic, turbulent period of transition — analogous to a forest recovering after a wildfire. Phase IV represented the new steady state: less diverse, but adapted to the conditions.”
The Membrane Fouling Story
Membrane fouling is one of the biggest operational headaches in any membrane bioreactor system. When the membrane clogs with organic matter, suspended solids, and microbial products, the transmembrane pressure (TMP) rises. When TMP hits a critical level, the membrane must be taken offline and cleaned, an expensive, time-consuming process.
In this study, membrane fouling behavior mirrored the microbial community dynamics almost exactly:
- Phases I & II: Stable fouling cycles of 6.9–13.5 days with manageable TMP increase rates (0.67 kPa/day slow, 11.8 kPa/day rapid).
- Phase III: Fouling rates spiked dramatically — 2.0 kPa/day in the slow phase and 24.4 kPa/day in the rapid phase. The membrane needed much more frequent cleaning.
- Phase IV: Fouling rates dropped to just 0.36 kPa/day in the slow phase, with cleaning cycles extending to 9.3–10.7 days — better even than baseline.
The explanation lies in the SMP. Stressed microbes secrete more SMP, proteins and polysaccharides that deposit on membrane surfaces and clog pores. Phase III’s crisis of SMP production (peaking at 178 mg/L) translated directly into accelerated membrane fouling. When the new, adapted community stabilized in Phase IV and SMP production normalized, so did membrane performance.
The molecular weight distribution of SMP also shifted critically in Phase III. The fraction of SMP particles in the 100 kDa to 0.45 μm size range particles that are perfectly sized to block the 0.1 μm membrane pores reached its highest proportion during this phase, contributing directly to the fouling crisis.
The Machine Learning Model: Teaching AI to Predict Water Quality
The final piece of this research revolves around the practical and exciting development of a machine learning model aimed at predicting effluent Chemical Oxygen Demand (COD) under varying salinity conditions. Biological wastewater treatment is inherently complex, with the relationship between input variables, such as salt concentration, organic load, temperature, and time, and output performance (effluent quality) being nonlinear and challenging to predict using simple equations. Machine learning is particularly adept at identifying patterns in intricate, multivariable data, making it a fitting choice for wastewater treatment prediction.
The researchers employed the Adaboost (Adaptive Boosting) algorithm, which combines multiple weak predictive models into a single, robust model. This approach is analogous to consulting a committee of 100 advisors, each with specialized knowledge, whereby weighing their votes leads to a more accurate decision than relying on any single advisor. Adaboost is advantageous compared to a single decision tree, which may overfit training data, and a random forest, which can be computationally intensive; it generally provides higher accuracy with a lower risk of overfitting, thus generalizing better to unseen data.
The model was designed with practical and measurable inputs that real wastewater treatment plants would routinely monitor, including influent COD concentration, NaCl concentration, operational phase, and time of exposure at each salinity level. The output focused on effluent COD, a key performance measure. Approximately 40 data points were collected during the experiment, with 70% used for training the model and 30% reserved for testing. The results revealed that the trained Adaboost model achieved an R² value of 0.9051 on the training set and 0.8027 on the test set, culminating in an overall R² of 0.8797. Notably, the average absolute prediction error was just 3.07%, indicating that the model can accurately predict effluent COD using easily measurable inputs regarding operating conditions.
The researchers are clear about the limitations: the model was built on data from one specific experimental setup, and it should not be extrapolated beyond the conditions studied without more data. But the concept is powerful. With more diverse training data from different AnMBR systems, salinities, and wastewater compositions, such models could become genuinely useful tools for plant operators.
The Future of AnMBR in Textile Treatment
This research points toward a future where AnMBR systems, perhaps enhanced with optimized operational conditions, strategic additive dosing, and AI-powered monitoring, can reliably treat high-salinity textile wastewater at scale.
The ceramic membrane is a particularly promising component, with durability, chemical resistance, and compatibility with aggressive cleaning protocols make it well-suited to the harsh realities of industrial textile wastewater. As ceramic membrane costs continue to decline and manufacturing scales up, the economics of ceramic AnMBR systems will improve.
The combination of UASB’s proven track record in textile treatment with the added control offered by membrane filtration creates a compelling technology for both new installations and for refurbishing existing treatment plants. The UASB configuration is particularly attractive in this context because it can mitigate membrane fouling compared to fully mixed reactor designs — the upflow hydraulics create a less aggressive fouling environment at the membrane surface.
On the machine learning side, the next step is larger datasets. The current model was trained on ~40 observations from a single system. Future work incorporating data from multiple plants, different membrane types, varying temperatures, and a wider range of salinities could produce models robust enough for real-time adaptive process control — automatically adjusting aeration, flux, or chemical dosing based on predicted effluent quality.
Conclusion
At its core, this research is a story about adaptation. Salt, in sufficient concentration, is hostile to microbial life. But life — given enough time and the right selection pressure — finds a way. The microbial community that stabilized in Phase IV was different from the one that started the experiment, but it was functional. It was doing the job.
For engineers and scientists, the lesson is not to view high salinity as a permanent death sentence for biological treatment. Rather, it is a challenge to be managed: through careful acclimation, smart operational adjustments, enhanced monitoring of key indicators like SMP and TMP, and increasingly, through machine learning tools that can predict performance shifts before they become crises.
The textile industry is not going away. Its wastewater challenge is not going away either. But research like this brings us meaningfully closer to scalable, robust, economically viable solutions for one of industrial pollution’s toughest problems.
Reference
Wang, K., Zhang, H., Shen, Y., Li, J., Zhou, W., Song, H., Liu, M., & Wang, H. (2023). Impact of salinity on anaerobic ceramic membrane bioreactor for textile wastewater treatment: Process performance, membrane fouling and machine learning models. Journal of Environmental Management, 345, 118717. https://doi.org/10.1016/j.jenvman.2023.118717